sculptor
Valued Senior Member
We have been blessed by a strong polar vortex this winter which has kept mid latitude winter temperatures mild.
https://earth.nullschool.net/#curre...thographic=-89.60,81.75,381/loc=53.607,89.749
Unfortunately:
That has led to a hole in the ozone layer over the arctic
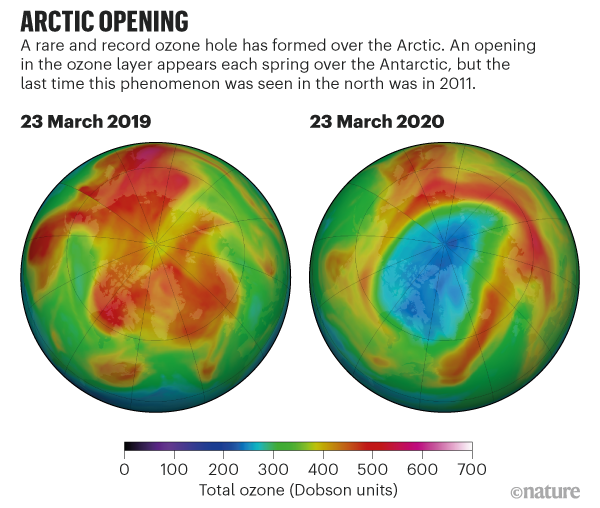
Cold temperatures and a strong polar vortex allowed chemicals to gnaw away at the protective ozone layer in the north.
There was more cold air above the Arctic than in any winter recorded since 1979, says Markus Rex, an atmospheric scientist at the Alfred Wegener Institute in Potsdam, Germany.
In the chilly temperatures, the high-altitude clouds formed, and the ozone-destroying reactions began.
How about that?
https://earth.nullschool.net/#curre...thographic=-89.60,81.75,381/loc=53.607,89.749
Unfortunately:
That has led to a hole in the ozone layer over the arctic

Cold temperatures and a strong polar vortex allowed chemicals to gnaw away at the protective ozone layer in the north.
There was more cold air above the Arctic than in any winter recorded since 1979, says Markus Rex, an atmospheric scientist at the Alfred Wegener Institute in Potsdam, Germany.
In the chilly temperatures, the high-altitude clouds formed, and the ozone-destroying reactions began.
How about that?
